DEVELOP
We partner with qualified vaccine manufacturers (usually from developing countries) to develop vaccines. The details of each partnership is manufacturer-specific, but in all cases we provide technology transfer and technical and financial support for process development and scale-up to clinical development, registration, and WHO pre-qualification.

Our partnerships are based on a common goal of making vaccines available for global public health. At the heart of product development partnerships is a commitment from the manufacturer to produce vaccines of high quality to meet WHO standards and to provide these vaccines to the public sector in sufficient quantities at an affordable price.
Process Development and Technology Transfer
Qualified manufacturing partners are identified through a careful selection process. Technology transfer is conditional upon the manufacturer producing vaccines of high-quality, at low-cost, in a country with a WHO-approved National Regulatory Authority (NRA).
To keep production costs down, IVI works to maximize the yield of vaccine antigens and to optimize antigen purification. By keeping manufacturing costs down, we make vaccines more affordable for developing countries and increase the probability of success in technology transfer of the vaccines. We also conduct quality control assays to ensure that the process is controlled and consistent from batch to batch and that the final product meets a pre-specified standard of quality.
| Tech transfer partner | Product Development Status |
|---|---|
| VaBiotech, Vietnam | Bivalent killed whole-cell oral cholera vaccine Partnered with VaBiotech on modifying the vaccine and tech-transferred quality control assays and reagents; vaccine currently licensed and used in Vietnam as mORC-VAX. |
| Shantha Biotechnics, India | Bivalent killed whole-cell oral cholera vaccine Partnered with Shantha on clinical development, licensure in India, and WHO prequalification; Shanchol™ WHO-prequalified in 2011 and currently used in the WHO stockpile. |
| EuBiologics, South Korea | Bivalent killed whole-cell oral cholera vaccine Worked with EuBiologics on late-stage clinical development; Euvichol® was WHO-prequalified in 2016; and Euvichol-Plus®, a plastic vial presentation, was prequalified in August 2017. |
| SK Chemicals, South Korea | Typhoid conjugate vaccine (Vi-DT) Phase I trial in the Philippines is completed (published), Phase II trial in the Philippines is on going, Phase III is being prepared; Chemistry, Manufacturing, and Controls (CMC) plan for Phase III agreed upon with BMGF; Vaccine expected to be WHO-prequalified by 2022. |
| PT BioFarma, Indonesia | Typhoid conjugate vaccine (Vi-DT) Currently working with BioFarma on clinical development; Phase I completed in Indonesia (publication accepted); Phase II began in July 2018; Vaccine expected to be licensed in Indonesia in 2022; WHO-prequalification plan under discussion. |
| Incepta Vaccine Ltd., Bangladesh | Bivalent killed whole-cell oral cholera vaccine Typhoid conjugate vaccine (Vi- DT) Currently working with Incepta on clinical development and licensure of cholera vaccine for the domestic market. |
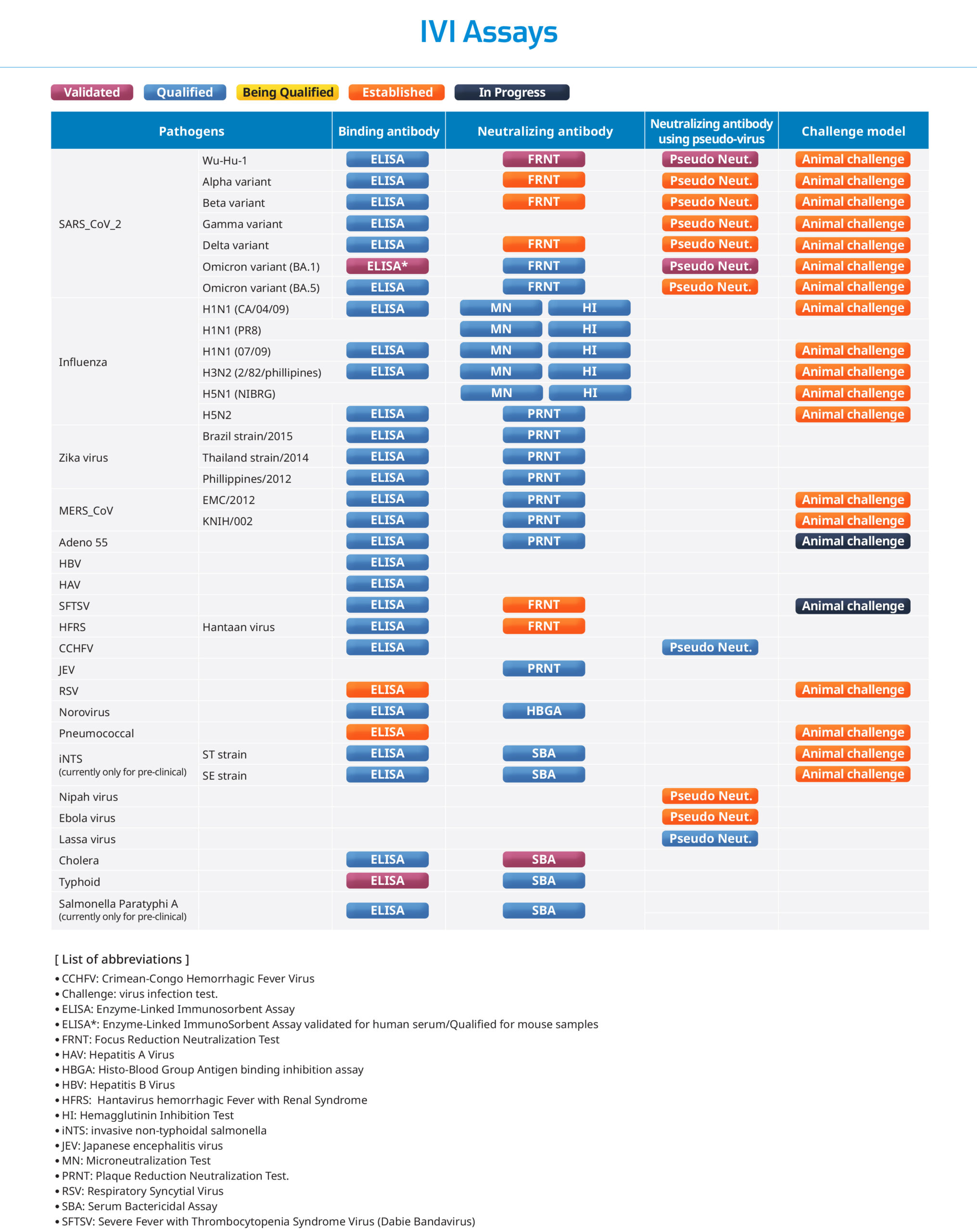
IVI Assays
IVI established in vitro and in vivo assays to assess humoral immune responses as well as protective effect of various viral and bacterial vaccines. The established assays are being used to support pre-clinical and clinical studies. Recently, the qualified ELISA and validated FRNT for SARS-CoV-2 are being used to asses binding antibody (IgG) and neutralizing antibody (nAb) level in COVID-19 vaccine clinical studies, respectively.
Clinical Development
To assess vaccine safety and efficacy in humans, we work with partners to plan and implement clinical trials (phases I to III, as well as phase IV). We provide training, technical assistance and financial support, as well as help with coordinating and monitoring trials to ensure that international standards and principles of good clinical practice are met.
Regulatory Review and Approval
We work with manufacturers and sponsors of new vaccine products throughout the regulatory approval process by helping them navigate the regulatory pathway and providing support regarding the various steps involved from preparation of the Investigational New Drug application, to inspection of the manufacturing facility. More specifically, we support manufacturers in getting their vaccine product approved by the WHO, which ensures that drugs and vaccines bought by countries and international procurement agencies (e.g., UNICEF) meet acceptable standards of quality, safety and efficacy.
Vaccine Safety
We also work to ensure vaccine safety by helping health authorities collect and analyze information from reports of adverse events that occur after administration of licensed vaccines. In collaboration with partners, we’ve developed a software tool known as the Vaccine Adverse Events Information Management System (VAEIMS), for collecting and processing information on adverse events following immunization (AEFI).
VAEIMS was created for the WHO Global Vaccine Initiative (GVSI), of which IVI is a participating partner, in order to efficiently transfer vaccine safety data from peripheral health care centers to a central database.
Following its pilot launch in Sri Lanka in 2015, the tool was rolled out in Chile, Iran, Lao PDR, Cambodia, Mongolia, and Vietnam. Based on positive feedback from participating countries, there are plans to expand VAEIMS to include an online version, a training program for NRAs, and the expansion of implementation in further countries.

