Cholera is an acute diarrheal infection caused by ingestion of food or water contaminated with the bacterium Vibrio cholerae. It affects both children and adults and can kill within hours if not properly treated. According to the WHO, there are 1.4 to 4.3 million cases and up to 143,000 cholera-related deaths annually; with a disproportionate burden in Africa and South Asia.
Improvements in water quality, hygiene and sanitation have been recognized as essential measures in preventing and controlling cholera. While effective, these are long-term measures and are challenging to implement in low-income countries with limited resources. Oral cholera vaccines can be an effective complementary tool that works in the short- to medium-term, especially during outbreaks when immediate action is needed.
Dukoral, a two-dose inactivated vaccine consisting of killed whole cells of Vibrio cholerae O1 and the B subunit of the cholera toxin, has been on the market since 1991. However, it is mainly used in developed countries as a traveler’s vaccine and has not been introduced in developing countries due to its relatively high cost.
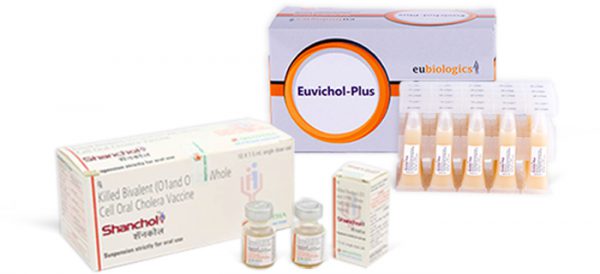
IVI developed a new low-cost two-dose killed whole-cell oral cholera vaccine. Development of the world’s first low-cost oral cholera vaccine was made possible due to an international public-private partnership led by IVI with partners from Sweden, Vietnam, India, Korea, and the U.S.
In the early 2000s, Vietnam had already developed and licensed its own oral cholera vaccine via a technology transfer from Sweden. However, in order for the vaccine to be used globally, it had to be modified to meet WHO requirements. In collaboration with VaBiotech, the Vietnamese manufacturer of the vaccine, IVI reformulated the vaccine and transferred the technology back to VaBiotech.
In addition, the technology was transferred to Shantha Biotechnics, a manufacturer in India – a country with a national regulatory authority (NRA) recognized by the WHO (a prerequisite for WHO prequalification). IVI worked with Shantha and other partners to perform clinical trials in Kolkata, India to demonstrate that the vaccine was safe and effective.
The vaccine was then licensed in India as Shanchol in 2009, and IVI worked closely with Shantha on WHO prequalification, which was obtained in 2011. This was a major achievement for both global health and IVI.
IVI recognized that more than one manufacturer was needed to ensure a reliable supply of the oral cholera vaccine for the global market. The Institute therefore started working with EuBiologics, a Korean company, in 2010 as its second manufacturing partner. IVI worked with Eubiologics to complete a technology transfer and implement the necessary trials for licensure in Korea and to obtain WHO prequalification.
Substantial progress has been made on the EuBiologics vaccine, Euvichol®. The vaccine was WHO-prequalified in 2015, and has since been sold through UNICEF to the global market. Additionally, Euvichol-Plus®, employing a user-friendly plastic container, achieved WHO prequalification in December 2017. The addition of Euvichol-Plus® reduced the price of the vaccine and has increased global supply to more than 25 million doses for 2018, with the potential to increase production further in the future.
More importantly, the extra capacity has contributed to reversing the cycle of low demand, low production, high price, and inequitable distribution, to one of: increased demand, increased production, reduced price, and increased access.
Meanwhile, a WHO stockpile using the oral cholera vaccine was established in late 2013, supported by Gavi, the Vaccine Alliance and is managed by the WHO and other partners. Initially the purpose of the 2 million dose stockpile was to expedite the delivery of cholera vaccines for rapid outbreak response in emergencies and crisis situations. With the availability of additional doses the stockpile now supports preventive campaigns in advance of an outbreak in communities at risk. From the initiation of the stockpile in 2013 through May 2018, around 25 million doses of OCV were deployed through campaigns in 19 different countries.
For years, OCV has been demonstrated to be safe and effective for cholera prevention and control in a number of countries worldwide. The vaccine is now one of the key tools of ‘Ending Cholera—A Global Roadmap to 2030,’ an ambitious strategy that was launched by the WHO in 2017, aimed at reducing cholera deaths by 90 percent by 2030.
While perceptions have been changing about using a vaccine to fight cholera, more evidence is still needed to demonstrate the impact of vaccination and to generate increased demand for OCV.
Currently, IVI continues to work on optimizing use of OCV in developing countries. For example, IVI recently published a study assessing whether the two-dose vaccine administered as a single dose provides sufficient protection against cholera. In addition, IVI recently started working with EuBiologics to obtain a Controlled Temperature Chain (CTC) label for Euvichol-Plus®, as the ability of OCVs to be kept outside traditional cold chain systems for limited periods is important for its use in many outbreak/reactive and emergency vaccination settings.
With the support of local partners, IVI introduced the oral cholera vaccine in Nepal, Ethiopia, Malawi and Mozambique, vaccinating more than 400,000 people at risk of cholera. The vaccine was introduced through campaigns in various settings (i.e.: a preventive campaign against endemic cholera in Ethiopia and Mozambique, and an emergency campaign against an outbreak in flood-stricken Malawi), showing that cholera vaccination is both practical and feasible.
Despite the progress made so far, IVI continues to work on cholera control, increasing vaccine supply, and advocating for the use of the vaccine – so that all at-risk communities are protected against cholera.
Learn more about IVI’s OCVs in the journal Vaccine: “The Euvichol story – Development and licensure of a safe, effective and affordable oral cholera vaccine through global public private partnerships”




