Your Excellency, Iván Duque Márquez, President of Colombia,
Honorable Madame, María Juliana Ruiz, First Lady of Colombia,
Honorable Madame, Kim Jung-sook, First Lady of the Republic of Korea,
Your Excellency, Fernando Ruiz, Minister of Health and Social Protection of Colombia,
Your Excellency, Juan Carlos Caiza Rosero, Ambassador of Colombia to the Republic of Korea,
Distinguished guests and friends,
It is with great pleasure that I welcome you, Mr. President and Madame First Lady, and your esteemed delegation to the International Vaccine Institute during your state visit to the Republic of Korea. It is also a great honor to receive Madame First Lady Kim Jung-sook, who is the Honorary President of the Korea Support Committee for IVI.
We were pleased to learn of the cooperation agreements between the governments of Colombia and South Korea, IVI’s host country, to strengthen health and vaccine development initiatives including knowledge sharing and capacity-building for infectious disease outbreak response.
In a historic year that has demonstrated the critical need for international partnership in protecting and strengthening global public health, investment in vaccine development and manufacturing capacity, and the open exchange of scientific research, we are particularly honored by your presence at IVI today.
Mr. President, allow me to take this opportunity to offer my sincere gratitude for your expression of intent to join IVI. The Memorandum of Understanding to be signed today between your Ministry of Health and Social Protection and IVI is a meaningful step toward what I believe will be a long and fruitful relationship.
Since its founding in 1997 as an initiative of the United Nations Development Programme, IVI has been working toward a vision of a world free of infectious diseases.
Our longstanding collaboration with Colombian researchers and institutions began in 2008 with the Dengue Vaccine Initiative, which was founded by IVI in partnership with the Sabin Vaccine Institute, Johns Hopkins University, the WHO, and the Gates Foundation to accelerate the development of dengue vaccines.
In Bogotá, DVI convened the Americas Dengue Prevention Board at the Instituto Nacional de Salud de Colombia to discuss strategies for dengue vaccine development and introduction. And in Medellín, DVI funded dengue surveillance studies which helped Colombia develop a mathematical model for understanding dengue transmission and the potential impact of vaccination.
Our newest and most significant collaboration focuses on the development and WHO-prequalification of a vaccine for chikungunya, a debilitating disease for which no approved vaccine or treatment is currently available. A WHO-prequalified chikungunya vaccine would have a significant impact on the lives of the millions of people in Latin America who are at risk of chikungunya infection.
Working with the Centro de Estudios en Infectología Pediátrica in Cali, and with collaborators in 4 other countries, IVI is leading a Phase 2/3 clinical trial of the chikungunya vaccine candidate developed by our partner Bharat Biotech. This initiative is funded
with more than 14 million US dollars from the Coalition for Epidemic Preparedness Innovations, the same organization that supported the development of the AstraZeneca and Moderna COVID-19 vaccines.
I am pleased to share that the first clinical trial participant received the chikungunya vaccine candidate during the late-stage trial in Costa Rica two weeks ago, and that the Colombian trial is on track to begin next month.
These initiatives are only a few examples of the types of partnerships that IVI fosters between governments, research institutions, academia, philanthropies, and other international organizations to advance sustainable solutions to urgent global health challenges.
IVI’s capabilities uniquely encompass the innovation and agility of a biotech company, the project portfolio of an academic research laboratory, and the leadership of an internationally recognized inter-governmental organization with core funding provided by the governments of the Republic of Korea, Sweden, India, and Finland.
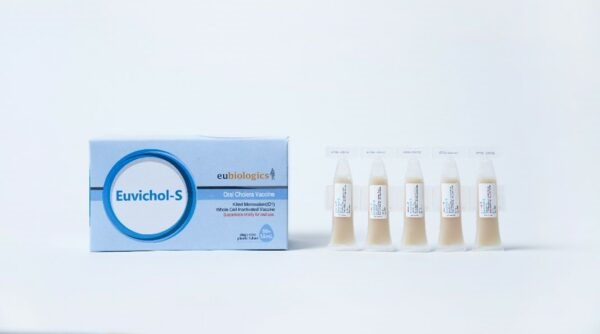
Through public-private partnerships, IVI developed the world’s first affordable oral cholera vaccine from discovery in the lab all the way through licensure and WHO prequalification, enabling the creation of a global stockpile managed by the WHO. To date, over 42 million doses of oral cholera vaccine have been deployed worldwide.
With the onset of the COVID-19 pandemic, IVI has also been working to advance COVID-19 vaccine candidates with over 20 collaborators at all stages of pre-clinical and clinical development, including the Republic of Korea’s SK bioscience’s vaccine candidate which is set to begin Phase 3 trials.
Mr. President, I once again extend a warm welcome to you and wish you success in your meeting with President Moon Jae-in, who has been an exceptional advocate for IVI in the international community along with Madame First Lady Kim Jung-sook.
Thank you, and I am looking forward very much to working closely with the people and government of Colombia.